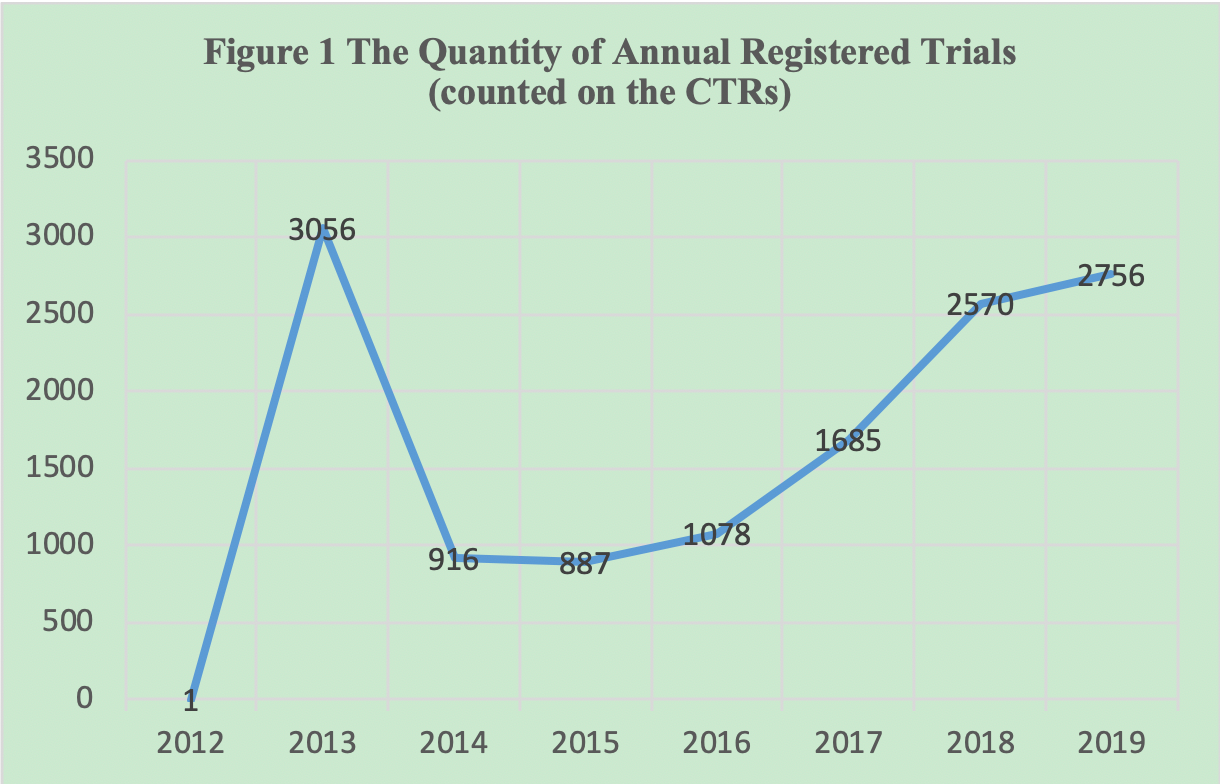
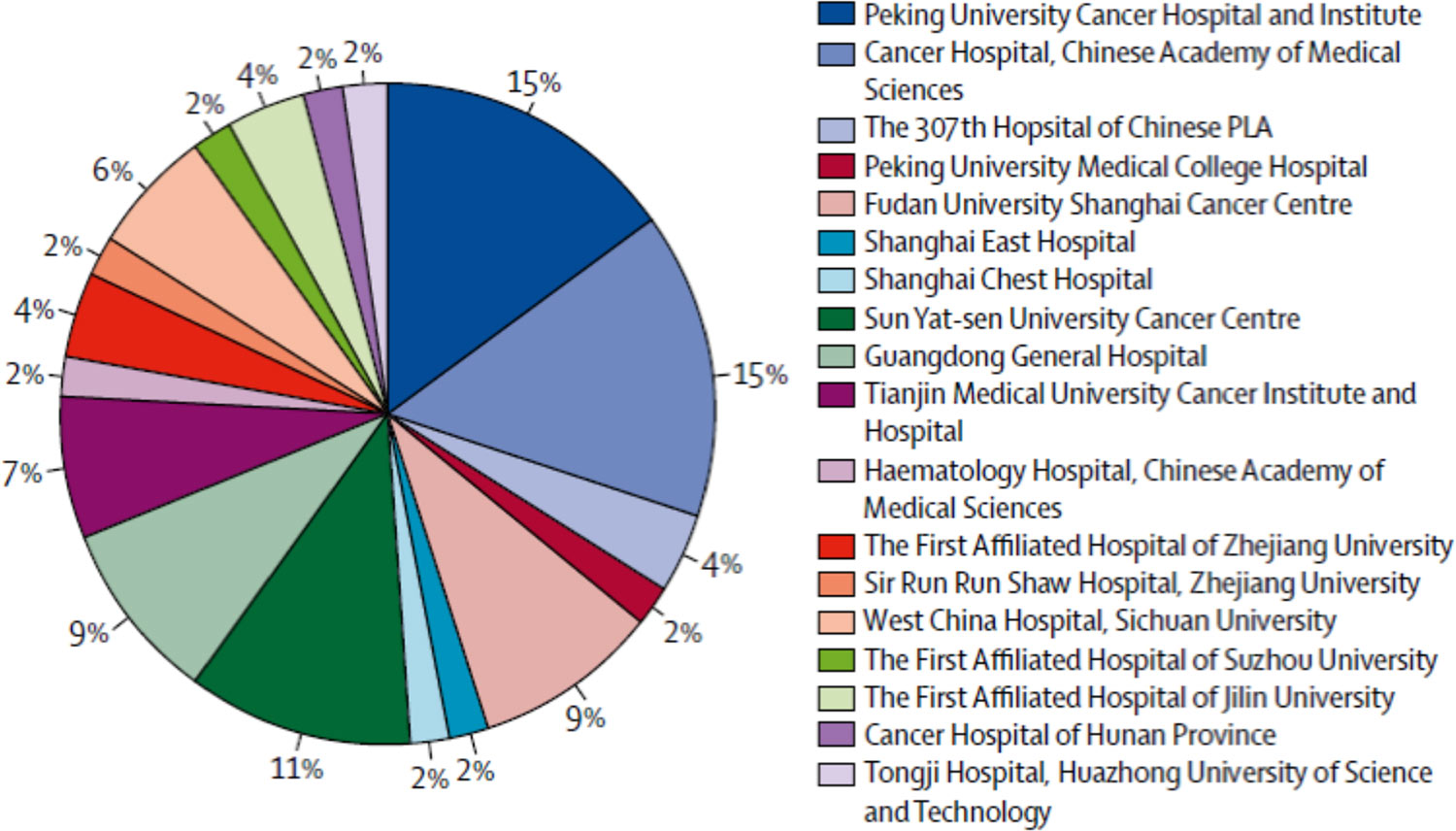
Regulatory Developments in China – Measures on Classification, Distribution, Manufacturing & Clinical Trial Management of Medical Devices - Compliance & Risks
Four key strategies for Western biotechs to expedite drug development in China - Clinical Trials Arena

NIH Launches “ClinRegs” Website Aggregating Country-By-Country Clinical Research Regulation – Policy & Medicine